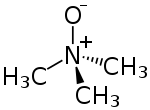
Trimethylamine N-oxide
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
N,N-Dimethylmethanamine N-oxide | |
| Other names
Trimethylamine oxide, TMAO, TMANO
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.013.341 |
| KEGG | |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C3H9NO | |
| Molar mass | 75.11 |
| Appearance | colorless solid |
| Melting point | 220 to 222 °C (428 to 432 °F; 493 to 495 K) (dihydrate: 96 °C) |
| good | |
| 5.4 D | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Trimethylamine N-oxide (TMAO) is an organic compound with the formula (CH3)3NO. It is in the class of amine oxides. Although the anhydrous compound is known, trimethylamine N-oxide is usually encountered as the dihydrate. Both the anhydrous and hydrated materials are white, water-soluble solids.
TMAO is found in the tissues of marine crustaceans and marine fish, where it prevents water pressure from distorting proteins and thus killing the animal. The concentration of TMAO increases with the depth at which the animal lives; TMAO is found in high concentrations in the deepest-living described fish species, Pseudoliparis swirei, which was found in the Mariana Trench, at a recorded depth of 8,076 m (26,496 ft).[1][2]
In animals, TMAO is a product of the oxidation of trimethylamine, a common metabolite of trimethyl quaternary ammonium compounds, like choline, trimethylglycine, and L-carnitine.[3] High TMAO concentrations are associated with an increased risk of all-cause mortality and cardiovascular disease.[4][5][6]
Marine animals
[edit]Trimethylamine N-oxide is an osmolyte found in molluscs, crustaceans, and all marine fishes and bony fishes. It is a protein stabilizer that serves to counteract the protein-destabilizing effects of pressure. In general, the bodies of animals living at great depths are adapted to high pressure environments by having pressure-resistant biomolecules and small organic molecules present in their cells, known as piezolytes, of which TMAO is the most abundant. These piezolytes give the proteins the flexibility they need to function properly under great pressure.[1][2][7][8][9]
TMAO decomposes to trimethylamine (TMA), which is the main odorant that is characteristic of degrading seafood.
TMAO in diet
[edit]TMAO levels increase with consumption of animal protein such as eggs, red meat, shellfish and total fish consumption.[10][11] Plant-based diets such as vegan, vegetarian and the Mediterranean diet lower TMAO levels.[11][12]
Chemistry
[edit]TMAO can be synthesized from TMA by treatment with hydrogen peroxide:[13]
- H2O2 + (CH3)3N → H2O + (CH3)3NO
The dihydrate is dehydrated by azeotropic distillation from dimethylformamide.[14]
Laboratory applications
[edit]Trimethylamine oxide is used in protein folding experiments to counteract the unfolding effects of urea.[15]
In the organometallic chemistry reaction of nucleophilic abstraction, Me3NO is employed as a decarbonylation agent according to the following stoichiometry:
- M(CO)n + Me3NO + L → M(CO)n−1L + Me3N + CO2
This reaction is used to decomplex organic ligands from metals, e.g. from (diene)Fe(CO)3.[13]
It is used in certain oxidation reactions, e.g. the conversion of alkyl iodides to the corresponding aldehyde.[16]
Effects on protein stability
[edit]The effects of TMAO on the backbone and charged residues of peptides are found to stabilize compact conformations,[17] whereas effects of TMAO on nonpolar residues lead to peptide swelling. This suggests competing mechanisms of TMAO on proteins, which accounts for hydrophobic swelling, backbone collapse, and stabilization of charge-charge interactions. These mechanisms are observed in Trp cage.[18]
Disorders
[edit]Trimethylaminuria
[edit]Trimethylaminuria is a rare defect in the production of the enzyme flavin-containing monooxygenase 3 (FMO3).[19][20] Those suffering from trimethylaminuria are unable to convert choline-derived trimethylamine into trimethylamine oxide. Trimethylamine then accumulates and is released in the person's sweat, urine, and breath, giving off a strong fishy odor.
Health effects
[edit]Mortality
[edit]High circulating TMAO concentrations are associated with an increased risk of all-cause mortality.[4][21]
Cardiovascular disease
[edit]High circulating TMAO concentrations are associated with an increased risk of cardiovascular events[4][21] and strokes in particular.[22]
Hypertension
[edit]High circulating TMAO concentrations are associated with an increased risk of hypertension.[23][24]
Potential toxicity
[edit]Exposure limit guidelines with a detailed description of toxicity are available such as "Recommendation from the Scientific Committee on Occupational Exposure Limits" by the European Union Commission.[25]
See also
[edit]References
[edit]- ^ a b Linley, T.D., M.E. Gerringer, P.H. Yancey, J.C. Drazen, C.L. Weinstock, A.J. Jamieson (2016). "Fishes of the hadal zone including new species, in situ observations and depth records of Liparidae". Deep Sea Research Part I: Oceanographic Research Papers. 114: 99–110. Bibcode:2016DSRI..114...99L. doi:10.1016/j.dsr.2016.05.003.
- ^ a b Gerringer, M.E., T.D. Linley, P.H. Yancey, A.J. Jamieson, E. Goetze, J.C. Drazen (2016). "Pseudoliparis swirei sp. nov.: A newly-discovered hadal snailfish (Scorpaeniformes: Liparidae) from the Mariana Trench". Zootaxa. 4358 (1): 161–177. doi:10.11646/zootaxa.4358.1.7. PMID 29245485.
- ^ Baker, J.R., Chaykin, S. (1 April 1962). "The biosynthesis of trimethylamine-N-oxide". J. Biol. Chem. 237 (4): 1309–13. doi:10.1016/S0021-9258(18)60325-4. PMID 13864146.
- ^ a b c Schiattarella GG, Sannino A, Toscano E, Giugliano G, Gargiulo G, Franzone A, Trimarco B, Esposito G, Perrino C. (2017). "Gut microbe-generated metabolite trimethylamine-N-oxide as cardiovascular risk biomarker: a systematic review and dose-response meta-analysis". European Heart Journal. 38 (39): 2948–2956. doi:10.1093/eurheartj/ehx342. PMID 29020409.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Li D, Lu Y, Yuan S, Cai X, He Y, Chen J, Wu Q, He D, Fang A, Bo Y, Song P, Bogaert D, Tsilidis K, Larsson SC, Yu H, Zhu H, Theodoratou E, Zhu Y, Li X. (2022). "Gut microbiota-derived metabolite trimethylamine-N-oxide and multiple health outcomes: an umbrella review and updated meta-analysis". Am J Clin Nutr. 116 (1): 230–243. doi:10.1093/ajcn/nqac074. PMC 9257469. PMID 35348578.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Dean YE, Rouzan SS, Loayza Pintado JJ, Talat NE, Mohamed ARH, Verma S, Anwar Kamdi Z, Gir D, Helmy A, Helmy Z, Afzal A, Mady T, Hazimeh Y, Aiash H. (2023). "Serum trimethylamine N-oxide levels among coronary artery disease and acute coronary syndrome patients: a systematic review and meta-analysis". Annals of Medicine and Surgery. 85 (12): 6123–6133. doi:10.1097/MS9.0000000000001426. PMC 10718322. PMID 38098555.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Yancey, P. (2005). "Organic osmolytes as compatible, metabolic, and counteracting cytoprotectants in high osmolarity and other stresses". J. Exp. Biol. 208 (15): 2819–2830. doi:10.1242/jeb.01730. PMID 16043587.
- ^ Velasquez M, Ramezani A, Manal A, Raj D (8 November 2016). "Trimethylamine N-Oxide: The good, the bad and the unknown". Toxins. 8 (11): 326. doi:10.3390/toxins8110326. PMC 5127123. PMID 27834801.
- ^ "What does it take to live at the bottom of the ocean?". BBC Earth. 2016. Archived from the original on 13 May 2016. Retrieved 19 May 2016.
- ^ Yang JJ, Shu XO, Herrington DM, Moore SC, Meyer KA, Ose J, Menni C, Palmer ND, Eliassen H, Harada S, Tzoulaki I, Zhu H, Albanes D, Wang TJ, Zheng W, Cai H, Ulrich CM, Guasch-Ferré M, Karaman I, Fornage M, Cai Q, Matthews CE, Wagenknecht LE, Elliott P, Gerszten RE, Yu D. (2021). "Circulating trimethylamine N-oxide in association with diet and cardiometabolic biomarkers: an international pooled analysis". The American Journal of Clinical Nutrition. 113 (5): 1145–1156. doi:10.1093/ajcn/nqaa430. hdl:10044/1/86226. PMC 8106754. PMID 33826706.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Lombardo M, Aulisa G, Marcon D, Rizzo G. (2022). "The Influence of Animal- or Plant-Based Diets on Blood and Urine Trimethylamine-N-Oxide (TMAO) Levels in Humans". Curr Nutr Rep. 11 (1): 56–68. doi:10.1007/s13668-021-00387-9. PMID 34990005.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Evans M, Dai L, Avesani CM, Kublickiene K, Stenvinkel P. (2023). "The dietary source of trimethylamine N-oxide and clinical outcomes: an unexpected liaison". Clin Kidney J. 16 (11): 1804–1812. doi:10.1093/ckj/sfad095. PMC 10616480. PMID 37915930.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b A. J. Pearson "Trimethylamine N-Oxide" in Encyclopedia of Reagents for Organic Synthesis, John Wiley & Sons, 2001: New York. doi:10.1002/047084289X.rt268
- ^ Soderquist, J. A., Anderson, C. L. (1986). "Crystalline anhydrous trimethylamine N-oxide". Tetrahedron Lett. 27 (34): 3961–3962. doi:10.1016/S0040-4039(00)84884-4.
- ^ Zou, Q., Bennion BJ, Daggett V, Murphy KP (2002). "The Molecular Mechanism of Stabilization of Proteins by TMAO and Its Ability to Counteract the Effects of Urea". J. Am. Chem. Soc. 124 (7): 1192–1202. doi:10.1021/ja004206b. PMID 11841287.
- ^ Volker Franzen (1973). "Octanal". Organic Syntheses; Collected Volumes, vol. 5, p. 872.
- ^ Shea JE, Feinstein SC, Lapointe NE, Larini L, Levine ZA (2015-03-03). "Regulation and aggregation of intrinsically disordered peptides". Proceedings of the National Academy of Sciences of the United States of America. 112 (9): 2758–2763. Bibcode:2015PNAS..112.2758L. doi:10.1073/pnas.1418155112. PMC 4352815. PMID 25691742.
- ^ Su Z, Mahmoudinobar F, Dias CL (2017). "Effects of Trimethylamine-N-oxide on the Conformation of Peptides and its Implications for Proteins". Physical Review Letters. 119 (10): 108102. Bibcode:2017PhRvL.119j8102S. doi:10.1103/physrevlett.119.108102. PMID 28949191.
- ^ Treacy, E.P., Akerman BR, Chow LM, Youil R, Bibeau C, Lin J, et al. (1998). "Mutations of the flavin-containing monooxygenase gene (FMO3) cause trimethylaminuria, a defect in detoxication". Human Molecular Genetics. 7 (5): 839–45. doi:10.1093/hmg/7.5.839. PMID 9536088.
- ^ Zschocke J, Kohlmueller D, Quak E, Meissner T, Hoffmann GF, Mayatepek E (1999). "Mild trimethylaminuria caused by common variants in FMO3 gene". Lancet. 354 (9181): 834–5. doi:10.1016/S0140-6736(99)80019-1. PMID 10485731. S2CID 9555588.
- ^ a b Guasti L, Galliazzo S, Molaro M, Visconti E, Pennella B, Gaudio GV, Lupi A, Grandi AM, Squizzato A. (2021). "TMAO as a biomarker of cardiovascular events: a systematic review and meta-analysis". Intern Emerg Med. 16 (1): 201–207. doi:10.1007/s11739-020-02470-5. PMID 32779113. S2CID 221099557.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Zhang H, Yao G. (2023). "Significant correlation between the gut microbiota-derived metabolite trimethylamine-N-oxide and the risk of stroke: evidence based on 23 observational studies". European Journal of Clinical Nutrition. 77 (7): 731–740. doi:10.1038/s41430-022-01104-7. PMID 35468932. S2CID 248368447.
- ^ Ge X, Zheng L, Zhuang R, Yu P, Xu Z, Liu G, Xi X, Zhou X, Fan H. (2020). "The Gut Microbial Metabolite Trimethylamine N-Oxide and Hypertension Risk: A Systematic Review and Dose-Response Meta-analysis". Adv Nutr. 11 (1): 66–76. doi:10.1093/advances/nmz064. PMC 7442397. PMID 31269204.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Han JM, Guo L, Chen XH, Xie Q, Song XY, Ma YL. (2024). "Relationship between trimethylamine N-oxide and the risk of hypertension in patients with cardiovascular disease: A meta-analysis and dose-response relationship analysis". Medicine (Baltimore). 103 (1): e36784. doi:10.1097/MD.0000000000036784. PMC 10766215. PMID 38181288.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Directorate-General for Employment SA, Scientific Committee on Occupational Exposure Limits, Nielsen GD, Pospischil E, Johanson G, Klein CL, et al. (2017). SCOEL/REC/179 trimethylamine: recommendation from the Scientific Committee on Occupational Exposure Limits. Publications Office of the European Union. doi:10.2767/440659. ISBN 978-92-79-66627-8. Retrieved 2023-12-17.
