Hordenine
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
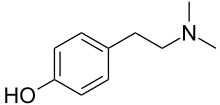
4-[2-(Dimethylamino)ethyl]phenol | |
| Other names
N,N-Dimethyltyramine; Peyocactin; Anhaline
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.007.920 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H15NO | |
| Molar mass | 165.236 g·mol−1 |
| Appearance | colorless solid |
| Melting point | 116 to 117 °C (241 to 243 °F; 389 to 390 K) |
| Boiling point | 173 °C (343 °F; 446 K) at 11 mm Hg; sublimes at 140–150 °C |
| high in: ethanol; ether; chloroform | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Hordenine is an alkaloid of the phenethylamine class that occurs naturally in a variety of plants, taking its name from one of the most common, barley (Hordeum species). Chemically, hordenine is the N-methyl derivative of N-methyltyramine, and the N,N-dimethyl derivative of the well-known biogenic amine tyramine, from which it is biosynthetically derived and with which it shares some pharmacological properties (see below). As of September 2012[update], hordenine is widely sold as an ingredient of nutritional supplements, with the claims that it is a stimulant of the central nervous system, and has the ability to promote weight loss by enhancing metabolism. In experimental animals, given sufficiently large doses parenterally (by injection), hordenine does produce an increase in blood pressure, as well as other disturbances of the cardiovascular, respiratory, and nervous systems. These effects are generally not reproduced by oral administration of the drug in test animals, and virtually no scientific reports of the effects of hordenine in human beings have been published.
Occurrence
[edit]The first report of the isolation from a natural source of the compound which is now known as hordenine was made by Arthur Heffter in 1894, who extracted this alkaloid from the cactus Anhalonium fissuratus (now reclassified as Ariocarpus fissuratus), naming it "anhalin".[1] Twelve years later, E. Léger independently isolated an alkaloid which he named hordenine from germinated barley (Hordeum vulgare) seeds.[2] Ernst Späth subsequently showed that these alkaloids were identical and proposed the correct molecular structure for this substance, for which the name "hordenine" was ultimately retained.[3]
Hordenine is present in a fairly wide range of plants, notably amongst the cacti,[4] but has also been detected i n some algae and fungi.[5][6][7] It occurs in grasses, and is found at significantly high concentrations in the seedlings of cereals such as barley (Hordeum vulgare) (about 0.2%, or 2000 μg/g), proso millet (Panicum miliaceum) (about 0.2%), and sorghum (Sorghum vulgare) (about 0.1%).[6] Reti, in his 1953 review of naturally occurring phenethylamines, notes that the richest source of hordenine is the cactus Trichocereus candicans (now reclassified as Echinopsis candicans), which was found to contain 0.5–5.0% of the alkaloid.[8]
Since barley, via its conversion to malt, is used extensively in the production of beer, beer and malt have been examined by several groups of investigators for the presence of hordenine. Citing a 1965 study by McFarlane,[9] Poocharoen reported that beer contained ~ 12–24 mg/L, wort contained about 11–13 mg/L, and malt contained about 67 μg/g of hordenine.[10] The hordenine content of various malts and malt fractions was extensively studied by Poocharoen himself, who also provided a good coverage of related literature up to 1983. This researcher found a mean concentration of hordenine in raw barley[a] around 0.7 μg/g; in green malts (i.e. barley that had been soaked in water for 2 days then germinated for 4 days), the mean concentration was about 21 μg/g, and in kilned malts (i.e. green malts that had been heated in a kiln for 1–2 days), the mean concentration was about 28 μg/g. When only green malt roots were examined, their mean content of hordenine was roughly 3363 μg/g, whereas the mean level in kilned malt roots was around 4066 μg/g.[10]
In barley, hordenine levels reach a maximum within 5–11 days of germination, then slowly decrease until only traces remain after one month. Furthermore, hordenine is localized primarily in the roots.[11] In comparing literature values for hordenine concentrations in "barley" or barley "malt", therefore, consideration should be made of the age and parts of the plant being analyzed: the figure of about 2,000 μg/g cited in the review by Smith,[6] for example, is consistent with Poocharoen's [10] figures for the hordenine levels in the roots of malted barley, but not in "whole" malt, where his figures of 21-28 μg/g are more consistent with McFarlane's figure of about 67 μg/g.[9] However, a wide range of variability is seen; a study by Lovett and co-workers of 43 different barley lines found concentrations of hordenine in roots ranging from 1 to 2625 μg/g fresh weight. These workers concluded that hordenine production was not under significant genetic control, but much more susceptible to environmental factors such as light duration.[12]
Biosynthesis
[edit]Hordenine is biosynthesized by the stepwise N-methylation of tyramine, which is first converted to N-methyltyramine, and which, in turn is methylated to hordenine. The first step in this sequence is accomplished by the enzyme tyramine N-methyltransferase (tyramine methylpherase), but if the same enzyme is responsible for the second methylation that actually produces hordenine is uncertain.[11][13]
Chemistry
[edit]Basicity
[edit]Since the hordenine molecule contains both a basic (amine) and acidic (phenol) functional group, it is amphoteric.
The apparent (see original article for discussion) pKas for protonated hordenine are 9.78 (phenolic H) and 10.02 (ammonium H).[14]
Common salts are hordenine hydrochloride,[15] R-NH3+Cl−, m.p. 178 °C, and hordenine sulfate,[16] (R-NH3+)2SO42−, m.p. 211 °C.
The "methyl hordenine HCl" which is listed as an ingredient on the labels of some nutritional supplements is in all likelihood simply hordenine hydrochloride, since the "description" of "methyl hordenine HCl" given by virtually all bulk suppliers of this substance corresponds to that for hordenine hydrochloride (or possibly just hordenine).[17] Five regioisomeric compounds would correspond to the name "methyl hordenine HCl", if it were interpreted according to the rules of chemical nomenclature: α-methyl hordenine, β-methyl hordenine, 2-methyl hordenine, 3-methyl hordenine, and 4-O-methyl hordenine - each in the form of its HCl salt; N-methyl hordenine is better known as the natural product candicine, but is excluded from the possibilities because it is a quaternary ammonium salt that cannot be protonated, hence cannot form a hydrochloride salt.
Synthesis
[edit]The first synthesis of hordenine is due to Barger: 2-phenylethyl alcohol was first converted to 2-phenylethyl chloride using PCl5; this chloride was reacted with dimethylamine to form N,N-dimethyl-phenylethylamine, which was then nitrated using HNO3; the N,N-dimethyl-4-nitro-phenethylamine was reduced to N,N-dimethyl-4-amino-phenethylamine with Sn/HCl; this amine was finally converted to hordenine by diazotization/hydrolysis using NaNO2/H2SO4/H2O.[18]
A more efficient synthetic route was described by Chang and coworkers, who also provided references to earlier syntheses. This synthesis began with p-methoxy-phenylethyl alcohol, which was simultaneously O-demethylated and converted to the iodide by heating with HI; the resulting p-hydroxy-phenylethyl iodide was then heated with dimethylamine to give hordenine.[19]
Radio-labelled hordenine has been prepared by the hydrogenation of a mixture of 2-[14C]-tyramine and 40% formaldehyde in the presence of 10% Pd-on-charcoal catalyst. The labelled C in the hordenine is thus the C which is β- to the N.[20]
Hordenine labelled with 14C at the position α- to the N has also been prepared,[21]
Pharmacology
[edit]The first pharmacological study of hordenine to be recorded is that of Heffter, who was also the first to isolate it. Using the sulfate salt (see "Chemistry"), Heffter gave a subcutaneous dose of 0.3 g to a 2.8-kg cat (about 107 mg/kg), and observed no effects besides violent vomiting; the cat behaved normally within 45 mins. He also took a dose of 100 mg orally himself, without experiencing any observable effect. However, the alkaloid was observed to produce a paralysis of the nervous system in frogs.[1]
Working with Léger's (see "Occurrence") hordenine sulfate, Camus determined minimum lethal doses for the dog, rabbit, guinea pig, and rat (see "Toxicology"). The associated symptoms of toxicity following parenteral doses were: excitation, vomiting, respiratory difficulties, convulsions, and paralysis, with death occurring as a result of respiratory arrest.[22] In a subsequent paper, Camus reported that the intravenous (IV) administration of some hundreds of mg of hordenine sulfate to dogs or rabbits caused an increase in blood pressure and changes in the rhythm and force of contraction of the heart, noting also that the drug was not orally active.[23]
The cardiovascular and other effects of hordenine were reviewed in detail by Reitschel, writing in 1937.[24]
More modern studies were carried out by Frank and coworkers, who reported that IV administration of 2 mg/kg of hordenine to horses produced substantial respiratory distress, increased the rate of respiration by 250%, doubled the heart rate, and caused sweating without changes in basal body temperature or behavior. All effects disappeared within 30 mins. The same dose of hordenine given orally did not produce any of the effects seen after parenteral administration.[25]
In a 1995 study, Hapke and Strathmann reported that in dogs and rats, hordenine produced a positive inotropic effect on the heart (i.e. increased the strength of contraction), increased systolic and diastolic blood pressure, and increased the volume of peripheral blood flow. Movements of the gut were inhibited. Additional experiments on isolated tissue lead these investigators to conclude that hordenine was an indirectly acting adrenergic agent that produced its pharmacological effects by releasing stored norepinephrine (NE).[26]
Hordenine was found to be a selective substrate for MAO-B, from rat liver, with Km = 479 μM, and Vmax = 128 nM/mg protein/h. It was not deaminated by MAO-A from rat intestinal epithelium.[27]
In contrast to tyramine, hordenine did not produce contraction of isolated rat vas deferens, but a 25 μM concentration of the drug did potentiate its response to submaximal doses of NE, and inhibited its response to tyramine. However, the response to NE of isolated vas deferens taken from rats chronically treated with guanethidine was not affected by hordenine. The investigators concluded that hordenine acted as an inhibitor of NE reuptake in rat vas deferens.[27]
Hordenine has been found to be a potent stimulant of gastrin release in the rat, being essentially equipotent with N-methyltyramine: 83 nM/kg of hordenine (corresponding to about 14 mg/kg of the free base) enhancing gastrin release by roughly 60%.[28]
In a study of the effects of a large number of compounds on a rat trace amine receptor (rTAR1) expressed in HEK 293 cells, hordenine, at a concentration of 1 μM, had almost identical potency to that of the same concentration of β-phenethylamine in stimulating cAMP production through the rTAR1. The potency of tyramine in this receptor preparation was slightly higher than that of hordenine.[29]
Toxicology
[edit]LD50 in mice, by intraperitoneal (IP) administration: 299 mg/kg.[30] Other LD50 values given in the literature are: >100 mg/kg (mouse; IP),[31] as HCl salt: 113.5 mg/kg (mouse; route of administration unspecified)[32] Minimum lethal dose (as sulfate salt): 300 mg/kg (dog; IV); 2000 mg/kg (dog; oral); 250 mg/kg (rabbit; IV); 300 mg/kg (guinea pig; IV); 2000 mg/kg (guinea pig; subcutaneous); about 1000 mg/kg (rat; subcutaneous).[22]
From experiments aimed at identifying the toxin responsible for producing the locomotor disorder ("staggers") and rapidly lethal cardiac toxicosis ("sudden death") periodically observed in livestock feeding on the grass Phalaris aquatica, Australian researchers determined that the lowest doses of hordenine that would induce symptoms of "staggers" in sheep were 20 mg/kg IV, and 800 mg/kg orally. However, the cardiac symptoms of "sudden death" could not be evinced by hordenine.[33]
Although hordenine is capable of reacting with nitrosating agents (e.g. nitrite ion, NO2−) to form the carcinogen N-nitrosodimethylamine (NDMA), and was investigated as a possible precursor for the significant amounts of NDMA once found in beer,[10] it was eventually established that the levels of hordenine present in malt were too low to account for the observed levels of NDMA.[34]
Pharmacokinetics
[edit]The pharmacokinetics of hordenine have been studied in horses. After IV administration of the drug, the α-phase T1/2 was found to be about 3 mins., and the β-phase T1/2 was about 35 mins.[25]
Insect interactions
[edit]Hordenine has been found to act as a feeding deterrent to grasshoppers (Melanoplus bivittatus),[35] and to caterpillars of Heliothis virescens and Heliothis subflexa; the estimated concentration of hordenine that reduced feeding duration to 50% of control was 0.4M for H. virescens and 0.08M for H. subflexa.[36]
Plant interactions
[edit]Hordenine has some plant growth-inhibiting properties: Liu and Lovett reported that, at a concentration of 50 ppm, it reduced the radicle length in seedlings of white mustard (Sinapis alba) by around 7%; admixture with an equal amount of gramine markedly enhanced this inhibitory effect.[37]
See also
[edit]- Tyramine
- N-Methyltyramine
- Candicine
- N,N-Dimethyldopamine
- 4-Hydroxyamphetamine
- Pholedrine
- O-desmethylvenlafaxine
- Venlafaxine
Notes
[edit]References
[edit]- ^ a b Heffter, A. (1894). "Ueber Pellote". Arch. Exp. Pathol. Pharmakol. 34: 6586.
- ^ Léger, E. (1906). "Sur l'hordenine: alcaloide nouveau retiré des germes, dits touraillons, de l'orge". Compt. Rend. (in French). 142: 108–10.
- ^ Späth, E. (1919). "Über die Anhalonium-Alkaloide. I. Anhalin und Mezcalin". Monatshefte für Chemie (in German). 40 (2): 129–54. doi:10.1007/BF01524590. S2CID 104408477.
- ^ "Visionary Cactus Guide". erowid.org. Retrieved January 14, 2021.
- ^ Wheaton, T. A.; Stewart, I. (June 1970). "The distribution of tyramine, N-methyltyramine, hordenine, octopamine, and synephrine in higher plants". Lloydia. 33 (2): 244–54. PMID 5495514.
- ^ a b c Smith, T. A. (1977). "Phenethylamine and related compounds in plants". Phytochemistry. 16: 9–18. doi:10.1016/0031-9422(77)83004-5.
- ^ Lundström, Jan (1989). Chapter 2 β-Phenethylamines and Ephedrines of Plant Origin. The Alkaloids: Chemistry and Pharmacology. Vol. 35. pp. 77–154. doi:10.1016/S0099-9598(08)60123-6. ISBN 9780124695351.
- ^ Reti, L. (1953). Chapter 22 β-Phenethylamines. The Alkaloids: Chemistry and Physiology. Vol. 3. pp. 313–338. doi:10.1016/S1876-0813(08)60144-X. ISBN 9780124695030.
- ^ a b McFarlane, W. D. (1965). "Tyrosine derived amines and phenols in wort and beer". Proc. Europ. Brew. Conv.: 387.
- ^ a b c d e Poocharoen, Boonthong (1983). Determination of selected secondary and tertiary amine alkaloids in barley malt (Thesis). hdl:1957/27227.
- ^ a b Mann, Jay D.; Mudd, S. Harvey (January 1963). "Alkaloids and Plant Metabolism". Journal of Biological Chemistry. 238 (1): 381–385. doi:10.1016/S0021-9258(19)84008-5.
- ^ Lovett, John V.; Hoult, Anne H. C.; Christen, Olaf (August 1994). "Biologically active secondary metabolites of barley. IV. Hordenine production by different barley lines". Journal of Chemical Ecology. 20 (8): 1945–1954. doi:10.1007/BF02066235. PMID 24242721. S2CID 6435423.
- ^ "Tyrosine metabolism - Reference pathway". Kyoto Encyclopedia of Genes and Genomes – via genome.jp.
- ^ Kappe, Thomas; Armstrong, Marvin D. (May 1965). "Ultraviolet Absorption Spectra and Apparent Acidic Dissociation Constants of Some Phenolic Amines 1". Journal of Medicinal Chemistry. 8 (3): 368–374. doi:10.1021/jm00327a018. PMID 14323148.
- ^ CAS No. 6027-23-2
- ^ CAS No. 622-64-0
- ^ See, for example:http://www.alibaba.com/showroom/methyl-hordenine-hcl.html
- ^ Barger, George (1909). "CCXXXV.—Synthesis of hordenine, the alkaloid from barley". J. Chem. Soc., Trans. 95: 2193–2197. doi:10.1039/CT9099502193.
- ^ Cheng, Chao-Shing; Ferber, Claus; Bashford, Raymond I.; Grillot, Gerald F. (September 1951). "A New Synthesis of Hordenine and Other p-Dialkylaminoethylphenols and Some of Their Derivatives". Journal of the American Chemical Society. 73 (9): 4081–4084. doi:10.1021/ja01153a008.
- ^ Digenis, George A.; Burkett, J. W.; Mihranian, V. (April 1972). "A convenient synthesis of 2 - [14C] - hordenine". Journal of Labelled Compounds. 8 (2): 231–235. doi:10.1002/jlcr.2590080208.
- ^ Russo, C. A.; Gros, E. G. (August 1981). "Synthesis of 4-|2-(dimethylamino) ethyl-2-14C| phenol (hordenine-α-14C)". Journal of Labelled Compounds and Radiopharmaceuticals. 18 (8): 1185–1187. doi:10.1002/jlcr.2580180813.
- ^ a b L. Camus (1906). "L'hordénine, son degré de toxicité, symptômes de l'intoxication." Compt. Rend. 142 110-113.
- ^ L. Camus (1906), "Action de sulfate d'hordenine sur circulation." Compt. Rend. 142 237-239.
- ^ Rietschel, Hans Georg (1 March 1937). "Zur Pharmakologie des Hordenins" [On the pharmacology of hordenine]. Naunyn-Schmiedebergs Archiv für experimentelle Pathologie und Pharmakologie (in German). 186 (2): 387–408. doi:10.1007/BF01929674. S2CID 37359919.
- ^ a b Frank, M.; Weckman, T. J.; Wood, T.; Woods, W. E.; Tai, Chen L.; Chang, Shih-Ling; Ewing, A.; Blake, J. W.; Tobin, T. (November 1990). "Hordenine: pharmacology, pharmacokinetics and behavioural effects in the horse" (PDF). Equine Veterinary Journal. 22 (6): 437–441. doi:10.1111/j.2042-3306.1990.tb04312.x. PMID 2269269.
- ^ Hapke, HJ; Strathmann, W (June 1995). "Pharmakologische Wirkungen des Hordenin" [Pharmacological effects of hordenine]. Deutsche Tierärztliche Wochenschrift (in German). 102 (6): 228–232. OCLC 121700602. PMID 8582256.
- ^ a b Barwell, C J; Basma, A N; Lafi, M A K; Leake, L D (12 April 2011). "Deamination of hordenine by monoamine oxidase and its action on vasa deferentia of the rat". Journal of Pharmacy and Pharmacology. 41 (6): 421–423. doi:10.1111/j.2042-7158.1989.tb06492.x. PMID 2570842. S2CID 10301433.
- ^ Yokoo, Y.; Kohda, H.; Kusumoto, A.; Naoki, H.; Matsumoto, N.; Amachi, T.; Suwa, Y.; Fukazawa, H.; Ishida, H.; Tsuji, K.; Nukaya, H. (1 March 1999). "Isolation from beer and structural determination of a potent stimulant of gastrin release". Alcohol and Alcoholism. 34 (2): 161–168. doi:10.1093/alcalc/34.2.161. PMID 10344776.
- ^ Bunzow, James R.; Sonders, Mark S.; Arttamangkul, Seksiri; Harrison, Laura M.; Zhang, Ge; Quigley, Denise I.; Darland, Tristan; Suchland, Katherine L.; Pasumamula, Shailaja; Kennedy, James L.; Olson, Susan B.; Magenis, R. Ellen; Amara, Susan G.; Grandy, David K. (1 December 2001). "Amphetamine, 3,4-Methylenedioxymethamphetamine, Lysergic Acid Diethylamide, and Metabolites of the Catecholamine Neurotransmitters Are Agonists of a Rat Trace Amine Receptor". Molecular Pharmacology. 60 (6): 1181–1188. doi:10.1124/mol.60.6.1181. PMID 11723224. S2CID 14140873.
- ^ Shinoda, Masato; Ohta, Setsuko; Takagi, Yoshinari (1977). "放射線障害防護薬剤に関する研究(第17報)フェネチラミン系化合物の放射線障害防護効力について" [Studies on Chemical Protectors against Radiation. XVII. Radioprotective Activities of Phenethylamine Compounds]. Yakugaku Zasshi (in Japanese). 97 (10): 1117–1124. doi:10.1248/yakushi1947.97.10_1117. PMID 592104.
- ^ Batista, Leonia Maria; Almeida, R. Nobrega de (1997). "Central effects of the constituents of Mimosa opthalmocentra Mart. ex Benth" (PDF). Acta Farmaceutica Bonaerense. 16 (2): 83–86.
- ^ Merck Index (10th ed.). Rahway, New Jersey: Merck & Co. 1983. p. 687.
- ^ Bourke, Ca; Carrigan, Mj; Dixon, Rj (July 1988). "Experimental evidence that tryptamine alkaloids do not cause Phalaris aquatica sudden death syndrome in sheep". Australian Veterinary Journal. 65 (7): 218–220. doi:10.1111/j.1751-0813.1988.tb14462.x. PMID 3421887.
- ^ Poocharoen, Boonthong.; Barbour, James F.; Libbey, Leonard M.; Scanlan, Richard A. (November 1992). "Precursors of N-nitrosodimethylamine in malted barley. 1. Determination of hordenine and gramine". Journal of Agricultural and Food Chemistry. 40 (11): 2216–2221. doi:10.1021/jf00023a033.
- ^ Harley, K. L. S.; Thorsteinson, A. J. (1 May 1967). "The influence of plant chemicals on the feeding behavior, development, and survival of the two-striped grasshopper, Melanoplus bivittatus (Say), Acrididae: Orthoptera". Canadian Journal of Zoology. 45 (3): 305–319. doi:10.1139/z67-043.
- ^ Bernays, E. A.; Oppenheim, S.; Chapman, R. F.; Kwon, H.; Gould, F. (1 February 2000). "Taste Sensitivity of Insect Herbivores to Deterrents is Greater in Specialists Than in Generalists: A Behavioral Test of the Hypothesis with Two Closely Related Caterpillars". Journal of Chemical Ecology. 26 (2): 547–563. doi:10.1023/A:1005430010314. S2CID 5695174.
- ^ Liu, D. L.; Lovett, J. V. (October 1993). "Biologically active secondary metabolites of barley. II. Phytotoxicity of barley allelochemicals". Journal of Chemical Ecology. 19 (10): 2231–2244. doi:10.1007/BF00979660. PMID 24248572. S2CID 8193525.
