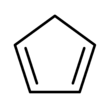
Cyclopentadiene
| |||

| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Cyclopenta-1,3-diene | |||
| Other names | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| Abbreviations | CPD, HCp | ||
| 471171 | |||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.008.033 | ||
| EC Number |
| ||
| 1311 | |||
| MeSH | 1,3-cyclopentadiene | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| Properties | |||
| C5H6 | |||
| Molar mass | 66.103 g·mol−1 | ||
| Appearance | Colourless liquid | ||
| Odor | irritating, terpene-like[1] | ||
| Density | 0.802 g cm−3 | ||
| Melting point | −90 °C; −130 °F; 183 K | ||
| Boiling point | 39 to 43 °C; 102 to 109 °F; 312 to 316 K | ||
| insoluble[1] | |||
| Vapor pressure | 400 mmHg (53 kPa)[1] | ||
| Acidity (pKa) | 16 | ||
| Conjugate base | Cyclopentadienyl anion | ||
| −44.5×10−6 cm3 mol−1 | |||
Refractive index (nD)
|
1.44 (at 20 °C)[3] | ||
| Structure | |||
| Planar[4] | |||
| 0.419 D[3] | |||
| Thermochemistry | |||
Heat capacity (C)
|
115.3 J·mol−1·K−1 | ||
Std molar
entropy (S⦵298) |
182.7 J·mol−1·K−1 | ||
Std enthalpy of
formation (ΔfH⦵298) |
105.9 kJ·mol−1[3] | ||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 25 °C (77 °F; 298 K) | ||
| 640 °C (1,184 °F; 913 K) | |||
| Lethal dose or concentration (LD, LC): | |||
LC50 (median concentration)
|
14,182 ppm (rat, 2 hr) 5091 ppm (mouse, 2 hr)[5] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 75 ppm (200 mg/m3)[1] | ||
REL (Recommended)
|
TWA 75 ppm (200 mg/m3)[1] | ||
IDLH (Immediate danger)
|
750 ppm[1] | ||
| Related compounds | |||
Related hydrocarbons
|
Benzene Cyclobutadiene Cyclopentene | ||
Related compounds
|
Dicyclopentadiene | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Cyclopentadiene is an organic compound with the formula C5H6.[6] It is often abbreviated CpH because the cyclopentadienyl anion is abbreviated Cp−.
This colorless liquid has a strong and unpleasant odor. At room temperature, this cyclic diene dimerizes over the course of hours to give dicyclopentadiene via a Diels–Alder reaction. This dimer can be restored by heating to give the monomer.
The compound is mainly used for the production of cyclopentene and its derivatives. It is popularly used as a precursor to the cyclopentadienyl anion (Cp−), an important ligand in cyclopentadienyl complexes in organometallic chemistry.[7]
Production and reactions[edit]

Cyclopentadiene production is usually not distinguished from dicyclopentadiene since they interconvert. They are obtained from coal tar (about 10–20 g/tonne) and by steam cracking of naphtha (about 14 kg/tonne).[8] To obtain cyclopentadiene monomer, commercial dicyclopentadiene is cracked by heating to around 180 °C. The monomer is collected by distillation, and used soon thereafter.[9] It advisable to use some form of fractionating column when doing this, to remove refluxing uncracked dimer.
Sigmatropic rearrangement[edit]
The hydrogen atoms in cyclopentadiene undergo rapid [1,5]-sigmatropic shifts. The hydride shift is however sufficiently slow at 0 °C to allow alkylated derivatives to be manipulated selectively.[10]

Even more fluxional are the derivatives C5H5E(CH3)3 (E = Si, Ge, Sn), wherein the heavier element migrates from carbon to carbon with a low activation barrier.
Diels–Alder reactions[edit]
Cyclopentadiene is a highly reactive diene in the Diels–Alder reaction because minimal distortion of the diene is required to achieve the envelope geometry of the transition state compared to other dienes.[11] Famously, cyclopentadiene dimerizes. The conversion occurs in hours at room temperature, but the monomer can be stored for days at −20 °C.[8]
Deprotonation[edit]
The compound is unusually acidic (pKa = 16) for a hydrocarbon, a fact explained by the high stability of the aromatic cyclopentadienyl anion, C
5H−
5. Deprotonation can be achieved with a variety of bases, typically sodium hydride, sodium metal, and butyl lithium. Salts of this anion are commercially available, including sodium cyclopentadienide and lithium cyclopentadienide. They are used to prepare cyclopentadienyl complexes.
Metallocene derivatives[edit]
Metallocenes and related cyclopentadienyl derivatives have been heavily investigated and represent a cornerstone of organometallic chemistry owing to their high stability. The first metallocene characterised, ferrocene, was prepared the way many other metallocenes are prepared: by combining alkali metal derivatives of the form MC5H5 with dihalides of the transition metals:[12] As typical example, nickelocene forms upon treating nickel(II) chloride with sodium cyclopentadienide in THF.[13]
- NiCl2 + 2 NaC5H5 → Ni(C5H5)2 + 2 NaCl
Organometallic complexes that include both the cyclopentadienyl anion and cyclopentadiene itself are known, one example of which is the rhodocene derivative produced from the rhodocene monomer in protic solvents.[14]
Organic synthesis[edit]
It was the starting material in Leo Paquette's 1982 synthesis of dodecahedrane.[15] The first step involved reductive dimerization of the molecule to give dihydrofulvalene, not simple addition to give dicyclopentadiene.

Uses[edit]
Aside from serving as a precursor to cyclopentadienyl-based catalysts, the main commercial application of cyclopentadiene is as a precursor to comonomers. Semi-hydrogenation gives cyclopentene. Diels–Alder reaction with butadiene gives ethylidene norbornene, a comonomer in the production of EPDM rubbers.
Derivatives[edit]

Cyclopentadiene can substitute one or more hydrogens, forming derivatives having covalent bonds:
- Bulky cyclopentadienes
- Calicene
- Cyclopentadienone
- Di-tert-butylcyclopentadiene
- Methylcyclopentadiene
- Pentamethylcyclopentadiene
- Pentacyanocyclopentadiene
Most of these substituted cyclopentadienes can also form anions, and join cyclopentadienyl complexes.
See also[edit]
References[edit]
- ^ Jump up to: a b c d e f g NIOSH Pocket Guide to Chemical Hazards. "#0170". National Institute for Occupational Safety and Health (NIOSH).
- ^ William M. Haynes (2016). CRC Handbook of Chemistry and Physics [Physical Constants of Organic Compounds]. Vol. 97. CRC Press/Taylor and Francis. p. 276 (3-138). ISBN 978-1498754286.
- ^ Jump up to: a b c William M. Haynes; David R. Lide; Thomas J. Bruno, eds. (2016). CRC handbook of chemistry and physics : a ready-reference book of chemical and physical data (2016-2017, 97th ed.). Boca Raton, Florida: CRC Press. ISBN 978-1-4987-5428-6. OCLC 930681942.
- ^ Faustov, Valery I.; Egorov, Mikhail P.; Nefedov, Oleg M.; Molin, Yuri N. (2000). "Ab initio G2 and DFT calculations on electron affinity of cyclopentadiene, silole, germole and their 2,3,4,5-tetraphenyl substituted analogs: structure, stability and EPR parameters of the radical anions". Phys. Chem. Chem. Phys. 2 (19): 4293–4297. Bibcode:2000PCCP....2.4293F. doi:10.1039/b005247g.
- ^ "Cyclopentadiene". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ LeRoy H. Scharpen and Victor W. Laurie (1965): "Structure of cyclopentadiene". The Journal of Chemical Physics, volume 43, issue 8, pages 2765-2766. doi:10.1063/1.1697207
- ^ Hartwig, J. F. (2010). Organotransition Metal Chemistry: From Bonding to Catalysis. New York, NY: University Science Books. ISBN 978-1-891389-53-5.
- ^ Jump up to: a b Hönicke, Dieter; Födisch, Ringo; Claus, Peter; Olson, Michael. "Cyclopentadiene and Cyclopentene". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a08_227. ISBN 978-3527306732.
- ^ Moffett, Robert Bruce (1962). "Cyclopentadiene and 3-Chlorocyclopentene". Organic Syntheses; Collected Volumes, vol. 4, p. 238.
- ^ Corey, E. J.; Weinshenker, N. M.; Schaaf, T. K.; Huber, W. (1969). "Stereo-controlled synthesis of prostaglandins F-2a and E-2 (dl)". Journal of the American Chemical Society. 91 (20): 5675–7. doi:10.1021/ja01048a062. PMID 5808505.
- ^ Levandowski, Brian; Houk, Ken (2015). "Theoretical Analysis of Reactivity Patterns in Diels–Alder Reactions of Cyclopentadiene, Cyclohexadiene, and Cycloheptadiene with Symmetrical and Unsymmetrical Dienophiles". J. Org. Chem. 80 (7): 3530–3537. doi:10.1021/acs.joc.5b00174. PMID 25741891.
- ^ Girolami, G. S.; Rauchfuss, T. B.; Angelici, R. J. (1999). Synthesis and Technique in Inorganic Chemistry. Mill Valley, CA: University Science Books. ISBN 0-935702-48-2.
- ^ Jolly, W. L. (1970). The Synthesis and Characterization of Inorganic Compounds. Englewood Cliffs, NJ: Prentice-Hall. ISBN 0-13-879932-6.
- ^ Kolle, U.; Grub, J. (1985). "Permethylmetallocene: 5. Reactions of Decamethylruthenium Cations". J. Organomet. Chem. 289 (1): 133–139. doi:10.1016/0022-328X(85)88034-7.
- ^ Paquette, L. A.; Wyvratt, M. J. (1974). "Domino Diels–Alder reactions. I. Applications to the rapid construction of polyfused cyclopentanoid systems". J. Am. Chem. Soc. 96 (14): 4671–4673. doi:10.1021/ja00821a052.
- ^ Reiners, Matthis; Ehrlich, Nico; Walter, Marc D. (2018). Synthesis of 1,3,5-Tri-tert-Butylcyclopenta-1,3-diene and Its Metal Complexes Na{1,2,4-(Me3C)3C5H2} and Mg{η5-1,2,4-(Me3C)3C5H2)2. Inorganic Syntheses. Vol. 37. p. 199. doi:10.1002/9781119477822.ch8. S2CID 105376454.